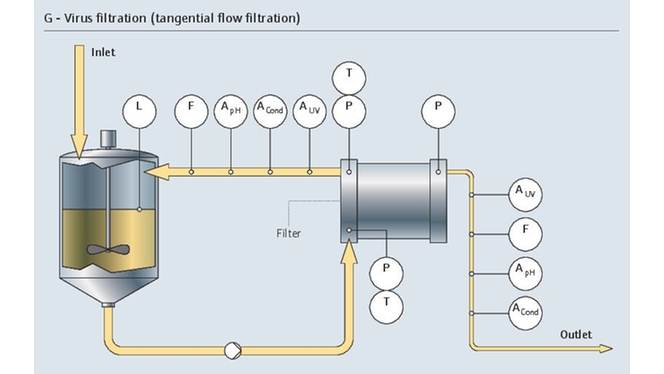
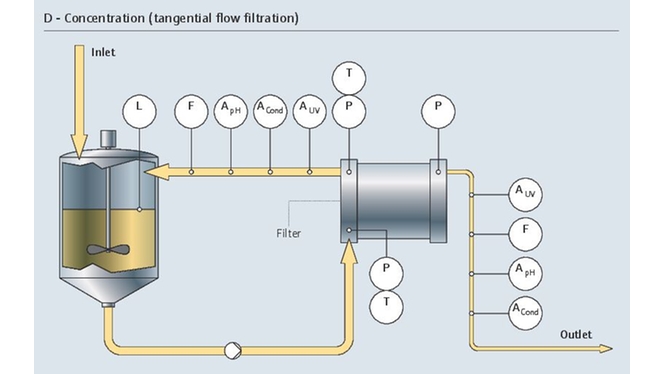
GMP pharmaceutical manufacturing transition from small scale process development can be challenging. Higher levels of automation and control, new instrumentation and technologies bring additional complexity. GMP compliance, industry standards and guidelines, all need to be addressed to ensure quality specifications are met. Our pharmaceutical manufacturing portfolio enables real-time monitoring and control of quality and process parameters while reaching productivity targets.
Key facts
100%
matching instruments
to user requirements - perfectly engineered, right first time
Learn how to standardize your instrumentation

Controlled fermentation
Involving living cells, fermentation is a very complex, sensitive and cost intensive bioprocess. Aseptic conditions must be strictly ensured throughout the biopharmaceutical manufacturing process. Consistent measurement is vital when meeting these requirements.
Our expertise in the field
Designed for long-term stability and accuracy, our instrumentation portfolio enables complete control of bio-reactor conditions:
- Rely on redundant pH and optical density measurement
- Enable consistent temperature control, optimized head space and foam level
- Monitor feed flow and pressure control

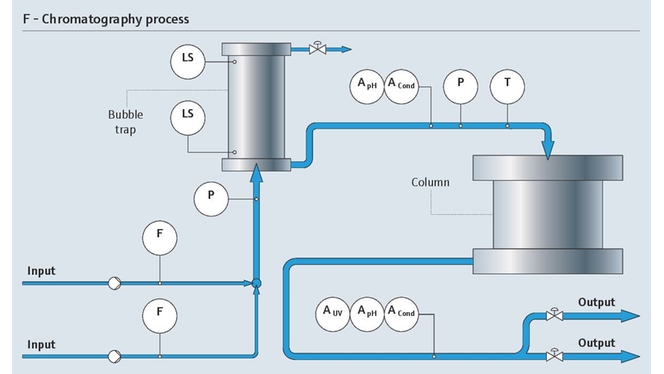
Efficient chromatography
Accurate measurement of conductivity and pH parameters is vital when leveraging chromatography. Moreover, detecting the target proteins to be separated from the complex mixture of the fermentation broth can be a complex challenge.
Our expertise in the field
Our monitoring solutions enable essential chromatography conditions to be maintained to support the separation process. We also offer UV process photometer sensors to facilitate the detection of target proteins:
- Drive pH and conductivity measurement in operation
- Optimize flow control
- Protect your process from over-pressure and over-temperature

Stable pH measurement
In a bioreactor pH is a critical control parameter. Each cell line has its optimum pH range for efficient cell growth and protein expression. From laboratory to large scale manufacturing, consistent and reliable data is required.
Our expertise in the field
The Endress+Hauser portfolio ensures stable inline pH measurement during fermentation for maximum reliability over all scales and allows an innovative calibration philosophy in the laboratory:
- Leverage Memosens cutting-edge sensor technology
- Deploy the same digital technologies in sensors and instrumentation from laboratory to large scale manufacturing
Benefits
Streamline your projects for the biopharmaceutical manufacturing process together with our employees, like Klaus Köhler. They provide both a complete range in instrumentation and profound industry expertise to find the best solution for your needs.
Key facts
80 %
reduction of instrument type diversity
Key facts
2 months
faster time to market
Key facts
0
changes of instruments, thanks to right-first-time specification
How we can help
Endress+Hauser offers a complete portfolio of scalable solutions for pharmaceutical manufacturing excellence. Our instrumentation and sensor devices enable accurate monitoring of critical process conditions, safeguarding compliance and guaranteeing batch release.
- Bioreactor and foam level measurement and control
- Protein separation in chromatography skids
- Analytical measurement in both laboratory and manufacturing
- Field instruments for easy calibration
- Entire instrumentation designed according to the ASME BPE standard